30
Apr
2024
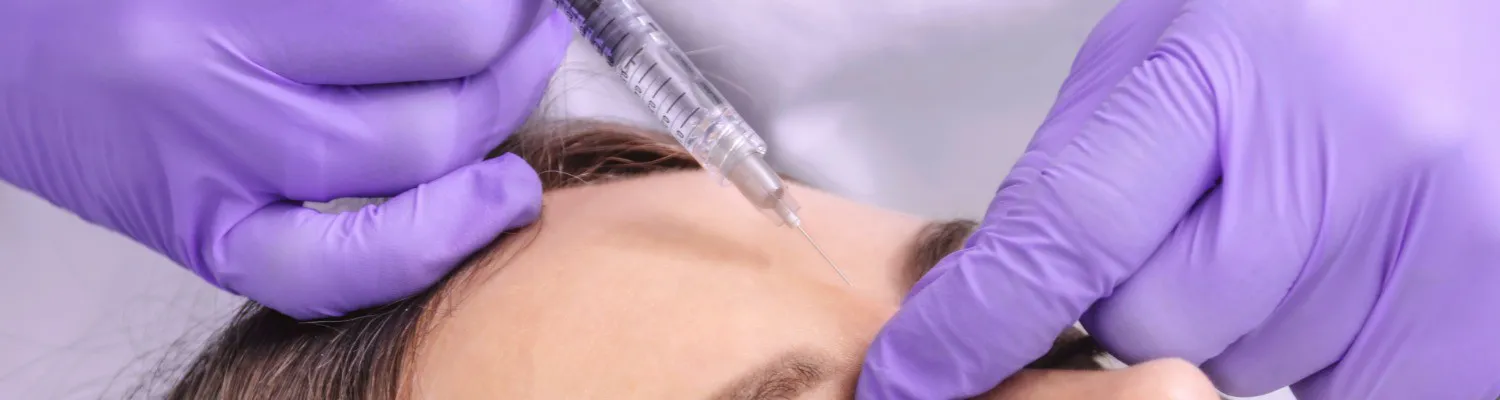
Rule 12.12 of the CAP Code expressly states that prescription-only medicines (POMs) or prescription-only medical treatments may not be advertised to the public. It is also an offence under the Human Medicines Regulations 2012 to in any way promote the prescription, supply, sale or use of a prescription-only product.
As with all sections of the CAP Code, rule 12.12 applies to ads in all media including websites and social media, and any reference to a POM is likely to amount to promotion of it.
A POM by any other name…
Stating the name of a particular POM, whether using a brand name like “Botox” or the generic form “botulinum toxin”, is always going to be a problem. The CAP Compliance team have issued numerous enforcement notices in relation to particular POMs (like Botox, Kenalog and POMs indicated for weight loss) which all make clear the fundamental notion that you simply can’t advertise POMs to the public.
Even if you don’t directly name the POM, an indirect reference is still likely to be a problem. For instance, with respect to weight-loss, we often see medicines presented as “skinny pens” or “skinny pills” which the CAP Compliance team has taken action against.
Some non-POM medicines may have a similar name to well-known POMs. For instance, Viagra Connect (which contains 50mg of sildenafil) was reclassified by the Medicines and Healthcare products Regulatory Agency (MHRA) as a ‘pharmacy medicine’ (P). This means it is available without prescription for use by men over 18 who have erectile dysfunction. There is no prohibition on the advertising of pharmacy medicines, which are included in the category referred to as ‘Over the Counter’ (OTC) medicines.
However, because other variations of Viagra, and other drugs used to treat erectile dysfunction, are currently still classified as POMs, advertisers should therefore take care that in promoting Viagra Connect, they do not indirectly promote POMs.
Does your treatment talk about treatments?
References to treating “excessive sweating” (hyperhidrosis) or any of the other conditions for which a POM is the only treatment, is likely to be considered an indirect promotion and therefore unacceptable.
However, provided no reference to the POM is made in ads, it may be acceptable to refer to a “consultation for hyperhidrosis”. Similarly, if a POM is only one of a range of effective treatments offered, it is more likely to be acceptable to refer to the condition.
Promote the consultation, not the POM.
It is often possible to talk about the services you offer more widely, but emphasis should usually be on promoting the consultation for particular conditions such as erectile dysfunction, excessive sweating, weight control issues or lines and wrinkles rather than promoting any named POMs which might be used to treat the condition.
Questions? The ASA Copy Advice team are on-hand to provide free and fast bespoke advice on your non-broadcast advertising.