10
Mar
2011
The long awaited report following the review into the Regulation of Cosmetic Interventions in England, led by NHS Medical Director, Sir Bruce Keogh and a panel of advisors has been published today calling for a complete reclassification of dermal filler products amongst other recommendations for the private cosmetic sector.
The Keogh committee also want to ensure that all practitioners are properly qualified for all the procedures that they offer, from cosmetic surgeons offering breast augmentations to those offering cosmetic injectable treatments. All of which would be overseen by an ombudsman to aid those who have received poor treatment. Thus far the report and its contents have been welcomed by key organisations within the aesthetic industry.
Commissioned by the Government following the PIP breast implant scandal, the review has looked at the products used for surgical and non-surgical procedures, the people who administer them, the way they are advertised and the advice and support patients and consumers are given. The report sets out how it would like to see the aesthetic and cosmetic sector better regulated, practitioners better trained and the public having proper redress should things go wrong.
On first dissecting the industry, Sir Keogh admitted to being surprised that non-surgical interventions, which can have major and irreversible impacts on health if something goes wrong, are almost entirely unregulated. He summed it up by saying; “In fact, a person having a non-surgical cosmetic intervention has no more protection and redress than someone buying a ballpoint pen or a toothbrush.”
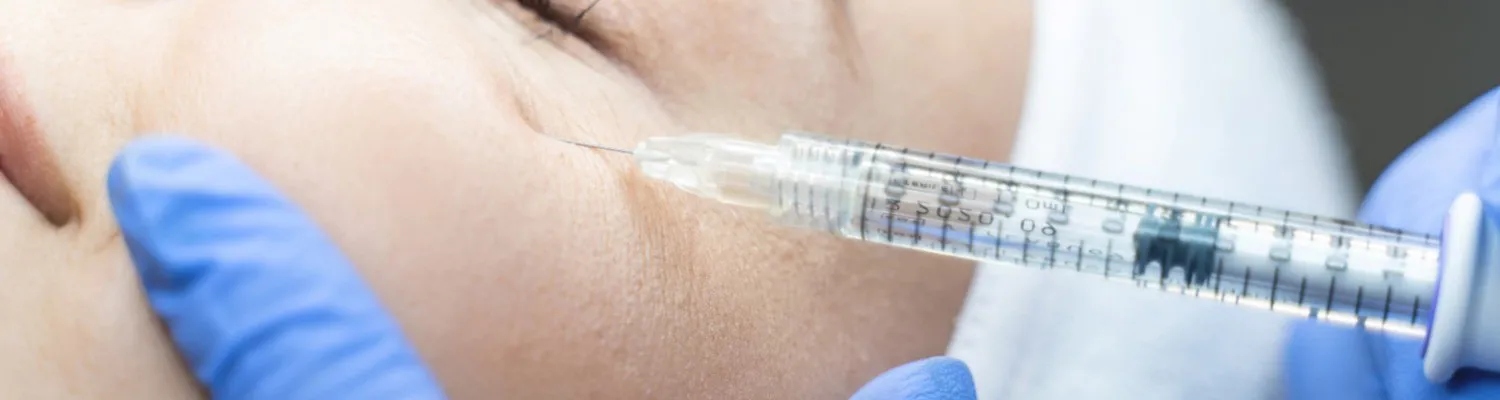
Of particular concern to the committee was the arena of dermal fillers where they noted that anyone can set themselves up as a practitioner, with no requirement for knowledge, training or previous experience and, in the absence of accredited training courses, anyone can set up a training course purporting to offer a (self-accredited) qualification. The casual use of products by unqualified individuals and reports of people buying injectable products over the Internet and self-administering alarmed the Keogh panel. Add to that the lack of sufficient checks in regard to the quality of the filler products themselves, currently poorly regulated under medical device regulations, which was likened to those imposed on a bottle of floor cleaner, meaning that the Review committee believes that dermal fillers are a crisis waiting to happen.
They also noted that data on rates of reported adverse reactions caused by dermal fillers and other non-surgical cosmetic interventions are poor because there are no formal reporting mechanisms in place for many of these products.
Sir Keogh raised the issue that previous attempts at self-regulation in the non-surgical side of the industry have failed, which he put down to them being largely voluntary codes which have meant that only the best in the industry commit themselves to better practice, whilst the unscrupulous and unsafe carry on as before. The report states that despite efforts from IHAS, Treatments You Can Trust has attained limited support from the sector, with concerns that, as a trade body, IHAS is not the appropriate organisation to run an independent register. It also points out that from a consumer perspective awareness of the register is low. It concludes that the failure of the sector to self-regulate may also partly reflect public attitudes which assume that there is already legislation. An observation which we would all be hard pressed to disagree with.
For this reason, one of the key recommendations of the report is to make dermal fillers prescription only medical devices to tackle both concerns head on. The committee are determined that the production of dermal fillers should fall under the same controls as other implants as this will ensure that only those fillers that have passed vigorous appraisals of safety will be available, and only those with ‘appropriate skill’ will be able to administer them.
Commenting on the report, Consulting Room™ Advisor, Consultant Plastic Surgeon and BAAPS President Rajiv Grover said; “We are thoroughly relieved that the Review has come to the same conclusions as we have over the years, specifically the urgent need for dermal fillers to require a prescription for use. This measure will kill three birds with one stone: regulating which ones come onto the market, who can inject them and automatically banning their advertising.”
The Review report notes that people undergoing non-surgical treatments should be able to be confident that their practitioner has the required skill and expertise to undertake the procedure successfully and safely.
They argue that the question is not ‘who should perform treatments’ but ‘what should adequate training and accreditation involve’? Noting that once the requirements for training are identified and understood, it should be possible to identify, for each professional group, which parts of the curriculum have been covered with prior training and which are consequently required to complete training. This will mean that different professional groups (e.g. medics like Doctors and Nurses versus non-medics like Beauty Therapists) will enter the training scheme at different points. Such a scheme could provide broad access, and may be able to provide professional training for those with no prior experience. The aim should be that, every practitioner, no matter what their starting point should attain the necessary skills and expertise to perform these varied procedures safely and to a high standard.
The training and accreditation process should ensure that practitioners are able to identify and manage complications of treatment and the curriculum and training requirements should be regularly reviewed to ensure that all practitioners are adequately trained in emerging procedures; this will involve regular retraining for those who wish to perform the latest treatments.
The committee believes that anyone prescribing fillers, or performing other potentially harmful non-surgical cosmetic procedures, should be accountable to a professional regulator.
The Review committee thus recommends that:
Following on from this the Review committee found that the current regulation of non-surgical providers is insufficient to adequately protect public health and safety.
Therefore they recommend that:
Those training to be non-surgical practitioners should have a clear understanding of the requirement to operate from safe premises, and the responsibilities involved. The training curriculum should include topics such as infection control, treatment room safety and adverse incident reporting. The code of conduct for those on the register should include an obligation to abide by certain clearly defined minimum standards for premises.

Sally Taber, Director of the Independent Healthcare Advisory Services and responsible for managing www.TreatmentsYouCanTrust.org.uk welcomed the Keogh Review and said; "The successful implementation of the recommendations on non-surgical procedures to ensure patient safety relies on two things. First, to end bad practice, the new qualification for cosmetic injectables must be underpinned by medical knowledge to ensure these medical treatments are administered safely. Second, it is important that these recommendations are executed swiftly to stop patients falling through the net. IHAS will be happy to utilise its existing framework and established industry guidelines to work constructively with the Government to expedite the process."
In relation to the products themselves, the Review report states that dermal fillers that do not claim to have a medical purpose (i.e. they are purely for cosmetic use) are exempt from EU medical device regulations and CE marking requirements before being able to be sold, thus products falling outside the medical devices regulations are only covered by the general provisions of the EU General Product Safety Directive. This maintains only a very general responsibility on distributors to place on the market (or supply) products that are safe in normal or reasonably foreseeable use. Furthermore, when a product such as a dermal filler is used as part of a ‘professional service’ it is currently exempt from the EU General Product Safety Directive. This means that in effect some dermal fillers used in cosmetic interventions in the UK are exempt from any product safety regulations! While this does not necessarily mean that the products are unsafe, it does mean that consumers and patients are reliant on manufacturers’ and providers’ own assessment of the safety of the product.
The Review report highlights that proposed revisions to the EU Medical Devices Directive are currently being discussed and may result in all dermal fillers and other implants for cosmetic use being classified as medical devices and therefore subject to the safety checks required for CE marking, but even if these changes are agreed, they are unlikely to be implemented before 2018, which Keogh believes leaves the public unprotected for too long. Manufacturers are currently not required to notify the MHRA that they are bringing medical devices or cosmetic implants to market in the UK and it is difficult therefore to get data on product use. According to the report, as a rough estimate, there are between 140 and 190 dermal filler products available in the UK (including CE marked and non-CE marked products), and there are no restrictions on who can purchase these products.
It therefore recommends that:
The 67 page report of course goes into much more detail about the various aspects of the cosmetic intervention market within England and its findings and recommendations. The Review Committee believes that the Government needs to establish a regulatory framework that encompasses the whole cosmetic sector, rather than the ‘piecemeal’ set up that it currently sees. This would employ a ‘clear, consistent and proportionate approach that is able to adapt to new developments’. The panel therefore hope that their recommendations, taken together, form a new legislative framework, demanded consistently by stakeholders and contributors to the evidence process that is proportionate to the potential risks of cosmetic interventions. The full report (Review of the Regulation of Cosmetic Interventions) highlights three key areas in which changes are needed within the whole industry:
1. high quality care with safe products, skilled practitioners and responsible providers;
2. an informed and empowered public to ensure people get accurate advice and that the vulnerable are protected;
3. and, accessible redress and resolution in case things go wrong.
The key recommendations are thus laid out as follows:
High Quality Care
An Informed and Empowered Public
Accessible Resolution and Redress
Sir Bruce is keen to put the minds of practitioner’s at rest by clarifying; “these recommendations are not about increasing bureaucracy but about putting everyone’s safety and wellbeing first.”
He went on to say; “We would like to see everyone who chooses to have any cosmetic procedure better protected. We would like to see people who carry out procedures trained to a high standard. We would like the public to feel confident they are going to be well looked after and, if things go wrong, that they will be supported. And ultimately, if someone needs to step in on the side of patients, we think there should be an ombudsman to do that. We very much hope that our report will lead to a safer environment for patients and this industry in the future.”
All we have to see now is if the present Government will implement any of these recommendations or simply ignore them. With many of them being a mirror of some of the concerns raised in the 2005 reports by the then Healthcare Commission, the Expert Group and the Chief Medical Officer, it is a little disheartening for many to see that the same problems and inadequacies persist close to a decade later.
We will be analysing the Keogh Review is more detail in our next Member Magazine, but would welcome your thoughts and comments on this lengthy blog, please let us know using the form below.
Updated 24th April 2013 - 11:30am
BAPRAS statement in response to the Department of Health’s Review of Regulation on Cosmetic Interventions:
Tim Goodacre, BAPRAS Chair of Professional Standards and a member of the Review’s Working Group on Training and Education in Cosmetic Surgery, said:
“...This is a strong series of recommendations and we are pleased that many of our priorities have been included. We hope they achieve Parliamentary approval and support quickly so that the detailed implementation work can begin and patients can be sure of high quality care and controlled outcomes at all times.”
Full response from BAPRAS to the Keogh recommendations.
Updated 24th April 2013 - 12:00pm
BABTAC response to Government recommendations regarding Cosmetic Interventions:
Carolyne Cross, Chair of BABTAC & CIBTAC says ;
"Whilst not all the changes are consistent with our own recommendations for cosmetic interventions, we are quietly confident that the outcome will work to benefit both the consumers and the industry practitioners. It is our belief that any changes should work to improve consumer safety and awareness, without creating a market monopoly and driving up prices. Our original submission to the Review and subsequent representations highlighted a number of the same recommendations and we are pleased that the Committee has taken a proactive and impartial approach. In terms of the impact on the beauty industry, we believe that there will be some significant changes in terms of regulations and qualifications, and as an organisation we have been driving for improved standards of training and regulation of our own industry providers. For therapists, it is likely that they will have to undertake additional training to become compliant, and whilst this has a cost implication for each salon, the outcome – better protected clients - will be worth the additional expenditure. We hope that these changes will serve to support and develop already reputable businesses, whilst undermining the status of ‘rogue traders’ who practice without due care and attention."
Full response from BABTAC to the Keogh recommendations.
Updated 24th April 2013 - 12:15pm
Allergan (maker of botulinum toxin brand Botoxâ and the Juvedermâ range of dermal fillers) response to Government recommendations regarding Cosmetic Interventions:
"Allergan is in favour of regulatory changes to classify all cosmetic implants including dermal fillers as medical devices and supports many of the other proposals such as calls to strengthen the existing European Medical Device Directive, plans to establish accredited training standards for health care professionals administering cosmetic implants, as well as steps to improve consultation and record keeping of patient treatments.
......
The report calls for a proposal to re-classify dermal fillers as prescription-only medical devices. This will require further thought to ensure the necessary legislation would bring about positive change in ensuring optimal patient outcomes. Specifically, any legislation change of this type will require significant time and could also mean that medical aesthetic nurses, who are generally already highly skilled and experienced, will need to re-train in order to obtain a prescribing licence."
Full response from Allergan to the Keogh recommendations.
Updated 24th April 2013 - 7:15pm
British College of Aesthetic Medicine (BCAM) response to Government recommendations regarding Cosmetic Interventions:
Dr Samantha Gammell, President BCAM said:
" BCAM is both pleased and relieved that the Review has put forward recommendations which are both sensible and proportionate in relation to cosmetic treatments. BCAM has long advocated the regulation of the use of dermal fillers in view of the potentially very serious complications which can arise from their use and we are delighted at the proposal that these products should become prescription only medicines. We also welcome the committee’s recommendations regarding requirements for verifiable training in the field of aesthetic medicine which is the central focus of the College.
In addition to the above the Review proposes that cosmetic treatments of all types should only be carried out by qualified clinical professionals who are prescribers or under the supervision of such professionals which would end the practice of administration of treatments by unqualified and untrained individuals performing treatments in unsuitable facilities.
We are thrilled that the Review has taken on board the views we expressed in our evidence to them late last year and that so many of our suggestions have been adopted by Professor Keogh and his team in the final report. It is vital for the sake of the profession that cosmetic surgery and aesthetic medicine is appropriately regulated and is performed only by those who are qualified, skilled and able to deal with the rare but potentially serious complications.
We look forward to working with the Department of Health, Health Education (England) and the other professional bodies in shaping the future direction of the speciality in the coming months and hope that the government will take Professor Keogh’s recommendations seriously and recognise the importance of regulation in this sector "
Updated 25th April 2013 - 10:30am
Royal College of Surgeons (RCS) response to Government recommendations regarding Cosmetic Interventions:
Professor Norman Williams, President of the RCS, said;
"We welcome this report and the recommendations to address the vacuum of regulation and standards that currently exists in cosmetic surgery. Over the past decade there have been numerous reports, a great deal of discussion but little action. The failure of PIP implants was a wake-up call that brought into sharp focus the desperate need for Government intervention to ensure commercial interests no longer take priority over patient safety. We call on Government to work with us to implement these recommendations without delay.
We support the committee’s recommendation that only doctors on a General Medical Council specialist register should perform cosmetic surgery and work within the scope of their specialty specific training. This is a start to addressing concerns about unqualified practitioners engaged in this work including some overseas surgeons operating in the UK. We welcome the opportunity to set clear standards for training and practice to ensure all surgeons are certified to the same standard irrespective of where they are trained.
We are also pleased that, as a priority, the review proposes a National Breast Implant Registry should be operational within 12 months. The College has long pressed for mandatory databases for all surgical implants to improve patient safety by keeping an audit trail of device failures and complications. We hope this registry will be rolled out across surgery so that all medical devices are covered."
Full response from RCS to the Keogh recommendations.
Updated 8th May 2013 - 10:00am
British Association of Cosmetic Nurses (BACN) response to Government recommendations regarding Cosmetic Interventions:
"If practitioners are to be licensed - who will license and how would it work? If there is to be accredited education - how will this be designed? what will the entry levels be? who will deliver and where will it be delivered? how will experienced practitioners demonstrate required standards? We strongly object to anyone other than nurses, doctors and dentists qualifying to undertake these procedures.
If Premises are to be licensed - who will undertake this? what lessons have we learned? We support initiation of the HEE, PHSO, CQC and MHRA in engagement and consultation with clinical experts in pushing forward the recommendations towards credible and workable legislation. The group of experts in this case MUST include clinicians with direct and significant experience in the practices examined. Nurses experienced in aesthetics must be included and the BACN fully commit to engaging in this necessary process.
The BACN supports the development of a mandatory national register of non-surgical licensed clinicians with practitioners demonstrating competency in the relevant non surgical speciality, subject to audit, assessment, appraisals and revalidation.
The Voluntary Register TYCT failed on a number of levels. TYCT did not reflect the challenges required of voluntary regulation in this area. They did not address the fundamentals of regulating individual providers. They were governed by a number of large organisations, driven by commercial imperatives who themselves demonstrated unacceptable standards which have been highlighted for change in the report. The practices of these TYCT Quality Marked companies were responsible for the initiation and basis of the report. The notion that "only the best commit to TYCT" does not stand up to examination. TYCT having failed to gain trust and credibility, despite its best efforts, should not have a place in the future regulatory landscape. We would advocate a new, robust, specifically tailored standards agency which is non-commercial, non-partisan and which draws on all current professional standards and evidence of good practice.
We agree strongly with the views expressed by the RCS that only trained clinicians should undertake these medical procedures and to advocate otherwise compromises public safety, which is the main issue that this report is designed to address.
The report appears to suggest that those who are not medical may become qualified to practice under supervision. With patient safety foremost, there is absolutely no imperative for this to be the case, and "under supervision " is open to interpretation.. In the NHS, Healthcare Assistants at the bedside rather than qualified nurses has proved this point.
Nurses and doctors are licensed and regulated by and accountable to their statutory bodies, The NMC and GMC. The BACN questions their level of engagement in the review and a lack of discussion on the responsibility they have to the registered nurses and doctors who opt to work in this specialty. The government committee believes that anyone performing potentially harmful non-surgical cosmetic treatments should be accountable to a professional regulator, such as NMC, GMC and GDC. We question why unregulated therapists are included in any plans for higher standards.
We agree that the scope of EU Medical Devices Directive should be extended to include dermal fillers. These devices should only be supplied to and administered by nurses, doctors and dentists who are educated, licensed, regulated and accountable. Prescription only medicines may be administered by ANYONE once prescribed.
We are engaging with other stakeholders - BCAM, BAD and manufacturers - to come together at the earliest opportunity to influence the future landscape. We will be lobbying the media and government."