Botox is one of the most recognised names in aesthetics, but it is also one of the most misunderstood. People often use “Botox” to mean any anti-wrinkle injection, even though it refers to a specific prescription medicine (botulinum toxin) used to relax targeted muscles for a temporary effect. It is also used beyond facial lines, including for concerns such as excessive sweating and certain medical conditions.
That mix of familiarity and confusion is exactly why careful understanding matters. Online advice can blur key differences between Botox and treatments like dermal fillers, oversimplify what “one, two, or three areas” means, and gloss over the fact that this is a prescription-only medicine in the UK. When you know what is clinically established, what varies person to person, and what should be assessed in consultation, it becomes much easier to compare information and make sensible choices.
In this guide, you’ll learn the essentials of how Botox works in the body, where it is commonly used, how long effects typically last, and what side effects and limitations to keep in mind. You’ll also find practical guidance on choosing a qualified provider, understanding prescribing and supervision, and preparing the right questions for a consultation.
Your Botox Questions
Real Questions from Real People - Answered
Straightforward answers to the questions people like you are asking right now about Botox.

Botox.
What are the prices please i have Epilepsy but it is controlled so wanted to know if you can still do it
To view all the Botox questions, please click here.
Or click here to ask your own question.
Find A Verified Clinic
Trusted Botox Experts, Local to You
Easily connect with qualified, verified professionals for safe, reliable treatment.


Akselberg Clinic
5 The Arcade , Hitchin, SG5 1EE
Welcome to Akselberg Aesthetics Our clinic is led by Dr Victoria Russell-Akselberg, an experienced practitioner in regenerative treatments with a background as an NHS surgeon.

Beauty Clinic Brazzini
185 Battersea High Street, Battersea Park, London, SW11 3JS
Beauty Clinic Brazzini is a boutique aesthetic clinic based in Battersea, London, specialising in advanced, non-surgical face and body treatments. Led by experienced practitioner Elisa Brazzini
To find a Botox clinic near you, please click here.
Botox Reviews
Real Experiences. Honest Feedback.
Read what others have to say about Botox before you decide.

Kelly B, Huddersfield
"Kin is great, she talks everything through & ensures she has understood what you're looking for. There's never any pressure to get more done & she ensures to work at your pace in trying new things to see the results as you go.
Also the most gentle botox I've ever had. I've had both botox (forehead & side of eyes) and lip filler done here over the last year or so."
Joanne Pitman, Wickham
"Hayley is so professional and knowing what a highly qualified nurse she is was the reason I went to her to try Botox for the first time. Lovely salon, Hayley made me feel comfortable and relaxed and the results were brilliant, felt 10 years younger!"
Torsie Morley, Margate
"I’m so blown away with my Botox. I explained to Piroska how I’ve had many attempts & always had a floppy eye lid after the Botox had settled in, she listened & the results are incredible. I’m so happy, I looked younger and less tired, and she uses the purest Botox which works so you see the results super quick, so if you’re like me & inpatient this is such a bonus. Each patient is different our faces are different, this is where Piroska stands out, she’s studied the face & understands this. Thank you so much. I’ve also had the B12 jabs, so good, if you’re feeling fatigue & low iron, I felt so energised I highly recommend this too especially if you’re a sporty person."
To view all the Botox reviews, please click here.
Botox Before And After Images
See the Results for Yourself
Explore real Botox before and after images to understand what’s possible.

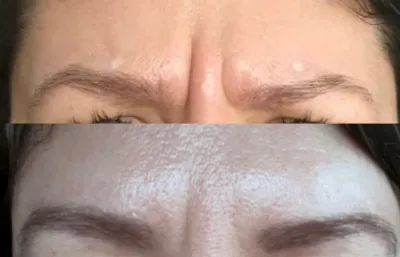
Anti-wrinkle treatment
Treatment of lines and wrinkles
Before And After Injectables
Before and after injectable treatments
To view the Botox gallery, please click here.
Botox Videos
Everything You’ve Been Wondering — Answered
See real products, expert advice, and patient stories brought to life.

The difference between Dermal Fillers and Botox treatment at Woodford Medical
Dr Louise Patterson discusses the key differences between Dermal Filler and Botox treatments in this informative video. Learn how each injectable treatment works and their unique benefits.
To view all the Botox videos, please click here.
What Is Botox and How Does It Work?
Botox is a popular cosmetic treatment but if you're considering getting it done and want to learn more, start here.

Botox is one of the most widely recognised names in medical aesthetics, yet it is also one of the most misunderstood. At its core, it refers to injections of botulinum toxin used to temporarily relax targeted muscles. This guide explains what Botox is, how it works in the body, and why it is used, without assuming prior knowledge or pushing you towards treatment.
Understanding Botox matters because decisions in this area are rarely simple. People often encounter mixed messages about safety, longevity, and how Botox compares with other injectable treatments such as dermal fillers. There is also understandable caution around the idea of using a prescription medicine for aesthetic purposes. Clear, evidence-aware information helps you separate what is clinically established from what varies by individual, practitioner, or treatment plan.
The full guide explores how Botox affects muscle activity, where it is commonly used, how long its effects typically last, and what safety and regulatory oversight look like in the UK. It also touches on limitations, side effects, and why practitioner expertise matters. Throughout, the focus is on informed choice and realistic expectations, grounded in how treatments are approached in clinical practice.
What Areas Can Be Treated With Botox?
When it comes to Botox treatment areas, there's no shortage of options to treat your lines and wrinkles. It's a perfect choice for men and women aiming to look more youthful, yet maintain a natural appearance.

Botox is often discussed as a single treatment, but in practice, it is used across a range of facial and non-facial areas, each with a different purpose and approach. This page explains which areas can be treated with Botox, and how those areas are typically defined in clinical practice, so you can make sense of what you may see online or hear in a consultation.
This matters because confusion around “areas” is common. Pricing, treatment plans, and expectations are often described in terms of one, two, or three areas, without always explaining what that means. At the same time, Botox is used not only for visible lines, but also for functional concerns such as excessive sweating or teeth grinding. Understanding the scope of treatment areas helps you compare information accurately and ask more informed questions.
The full guide explores common cosmetic treatment areas, medical and therapeutic uses, how areas are grouped in practice, and why treatment plans vary between individuals. It also explains where general information ends and where a professional consultation becomes essential.
Who Should You Trust To Do Your Botox?
To help you make safe decisions, we outline our top tips of things to consider before booking Botox with a trained medical professional.

Choosing who should carry out your Botox treatment is not a small decision, yet it is one many people find surprisingly difficult to navigate. This guide explains who can legally and professionally administer Botox in the UK and why those distinctions matter for safety, accountability, and informed care. It is designed for anyone considering treatment who wants clarity before committing time, money, or trust.
This topic matters because Botox is a prescription-only medicine, not a cosmetic product. That status affects who can assess you, who can prescribe the treatment, and how responsibility is managed if something does not go to plan. In practice, patients often encounter a wide mix of practitioners, titles, and clinic setups, all offering Botox under different models of care. Without a clear explanation, it can be hard to understand what is appropriate, what is expected, and what questions you should reasonably ask.
The full guide explores how Botox is regulated in the UK, the roles of doctors, dentists, nurses, and non-medical practitioners, and how prescribing and supervision should work in a responsible setting. It also outlines practical considerations that are commonly reviewed in clinical practice when assessing suitability and risk.
How MuchDoes Botox Cost?
If you're interested in Botox injections or want to find out the true cost of Botox treatment, please refer to this article for more information.

Understanding the cost of Botox is often one of the first and most confusing steps for anyone considering treatment. Prices can vary widely between clinics, and it is not always clear what those figures include or why they differ. This guide focuses on Botox pricing in the UK, explaining how costs are typically structured and what factors influence them, so you can make sense of the information you see before making any decisions.
Cost matters because Botox is a prescription-only medicine, and price should always be considered alongside safety, practitioner expertise, and clinical standards. Many people encounter online headline prices that are difficult to compare, particularly when clinics charge per area rather than per unit, or when consultation and review appointments are not clearly explained. Without context, it can be hard to judge whether a quote reflects appropriate care or simply a marketing tactic.
In the full guide, we explore typical starting prices, how clinics set their fees, what is usually included, and why unusually low prices can be a concern. We also look at practical questions to ask so you can compare clinics on a like-for-like basis.
This information is grounded in how Botox is assessed and prescribed in UK clinical practice, with an emphasis on clarity, regulation, and informed choice. If you want a calm, reliable starting point before reading further or speaking to a practitioner, this is where to begin.
Botox ForYour Frown or Glabella Lines
If you've got frown lines that appear when you furrow your brow (known medically as glabellar lines) Botox injections can definitely help to reduce the appearance of facial lines.

Botox for frown lines, also known as glabellar lines, is one of the most commonly discussed injectable treatments in the UK. These vertical lines form between the eyebrows and are closely linked to facial movement, muscle strength, and individual anatomy. While the treatment itself is widely recognised, understanding how and when it is used, and whether it is appropriate for you, is often less straightforward.
This topic matters because the frown line area sits at the centre of facial expression and near the eyes. Small differences in muscle activity, dosing, and placement can influence both safety and results. For many people researching Botox for the first time, it can be difficult to separate marketing language from clinical reality, or to know what questions are worth asking before booking a consultation.
The full guide explores how glabellar lines develop, how Botox is typically used in this area, what treatment planning involves, and what to consider around timing, longevity, cost, and practitioner choice. It also outlines known limitations and why individual assessment remains essential.
Botox ForCrow’s Feet
Crow's feet can be a sign that you've lived a happy life full of smiles, or that you've spent too much time in the sun. Fortunately, crow's feet are very treatable through Botox.

Botox for crow’s feet refers to the use of botulinum toxin injections to soften the fine lines that form at the outer corners of the eyes. These lines are closely linked to facial movement, particularly smiling and squinting, which is why they are often described as dynamic wrinkles. This page acts as a clear starting point for understanding how this treatment is used in clinical aesthetic practice, and what to consider before exploring it further.
Crow’s feet are a common concern, but they are also an area where people tend to feel cautious. The skin around the eyes is delicate, results can vary, and there is often confusion around what Botox can realistically do, how long the effects last, and how safety is managed. For many people, the challenge is not deciding whether they want treatment, but knowing how to assess it properly and what questions to ask first.
The full guide explores how crow’s feet develop, how botulinum toxin works in this area, what treatment typically involves, and how practitioners assess suitability and risk. It also outlines practical considerations such as treatment frequency, cost, and the importance of regulated care.
Botox ForBunny Lines
If the 'Bunny Lines' or 'Bunny Wrinkles' you get when you scrunch up your nose when doing certain facial expressions bother you, have you considered Botox?

Bunny lines are the fine creases that appear across the bridge and sides of the nose when you smile or scrunch your face. They are a common facial movement line, yet they are often misunderstood or mistaken for skin texture changes, which can make it difficult to determine whether treatment is appropriate or necessary.
This topic matters because bunny lines tend to become more noticeable gradually. Many people first notice them in photos or when other areas of the face have already been treated. That can raise practical questions, such as whether these lines respond to anti-wrinkle injections, how treatment differs from areas like the forehead, and what level of subtlety is realistic. Without clear information, it is easy to either overestimate what treatment can do or dismiss it without understanding the options.
The full guide explores what bunny lines are, how and why they form, how Botox is used in this area, what treatment typically involves, and the factors clinicians consider when assessing suitability. It also looks at limitations, potential risks, and why practitioner experience matters.
Rather than offering quick answers or outcomes, this page is designed to support informed conversations and realistic decision-making in a clinical context.
Botox ForForehead Lines
Wrinkles on your forehead are a sure sign of ageing and, unfortunately, only get worse as you get older. So, what can help? Botox!

Botox for forehead lines refers to the use of botulinum toxin injections to soften horizontal lines across the forehead caused by repeated muscle movement. It is one of the most commonly requested aesthetic treatments in the UK, but it is also one of the most frequently misunderstood. This page explains what the treatment involves and why careful assessment matters before deciding whether it is right for you.
Forehead lines can develop at different stages of life and for different reasons. Some only appear when the eyebrows are raised, while others become visible at rest over time. Because the forehead plays a central role in facial expression, treatment decisions here tend to raise specific concerns about safety, natural movement, and long-term planning. Understanding how Botox works in this area helps you compare options realistically and avoid assumptions based on marketing or anecdote.
The full guide explores how forehead lines form, how Botox is used in clinical practice, what factors affect dosing and placement, what results typically look like over time, and how costs, longevity, and side effects are usually discussed during consultation. It also explains why practitioner experience and assessment are especially important in this area.
Rather than offering promises or outcomes, this page focuses on how treatment decisions are typically assessed and reviewed in practice, helping you understand where general guidance ends and individual advice begins. If this feels relevant, the in-depth guide is the next step.
Botox ForA Dimpled Chin
If you're self-conscious about the dimpled appearance of your chin area, small injections of Botox in the chin muscle could make a big difference!
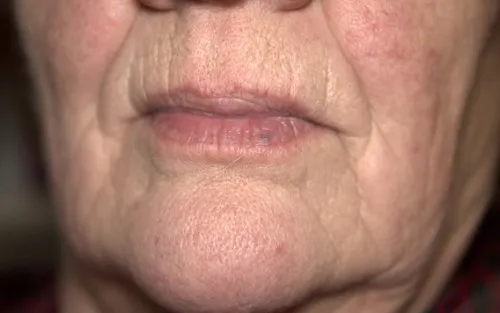
A dimpled or “orange peel” chin is a common lower-face concern, but it is often misunderstood. This guide explains how botulinum toxin injections are used to address chin dimpling caused by muscle activity, and why careful assessment matters before considering treatment. It is designed as a clear starting point for anyone researching options and wanting to understand whether this approach is relevant to them.
This topic matters because not all chin texture has the same cause. For some people, dimpling is related to the mentalis muscle pulling too strongly. For others, it is structural, genetic, or linked to skin quality rather than muscle movement. Without this distinction, it is easy to assume that a single treatment suits everyone, which is rarely the case in the lower face, where function and appearance are closely linked.
The full guide explores how practitioners assess chin movement, how injections are typically planned, what to expect from treatment timelines, how long effects may last, and which risks and limitations should be discussed in advance. It also outlines aftercare considerations and how costs are usually structured in the UK.
Throughout, the focus is on evidence-aware, clinical reasoning rather than trends or promises. This page will help you understand the principles and boundaries of treatment, while recognising that individual suitability is always confirmed in consultation.
Botox For Lip Lines
Nasolabial folds also known as lip lines, laugh lines or smile lines are caused by repeated facial expressions over time, Botox treatments can make a difference
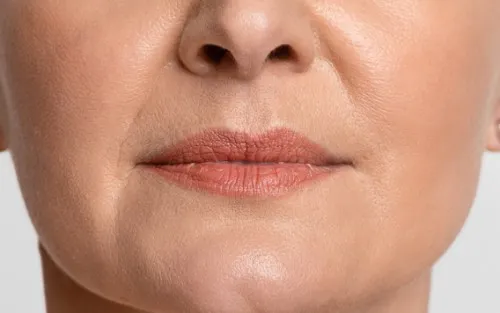
Botox for lip lines refers to the careful use of botulinum toxin to soften fine vertical lines around the mouth. These lines are often discussed alongside fillers or skin treatments, which can make it difficult to understand what Botox does and where it fits. This page explains the role of Botox specifically, in clear, practical terms, for readers who want to understand their options before taking things further.
Lip lines matter because the area around the mouth is both expressive and functional. Even subtle changes to muscle movement can affect how the lips feel and move, which is why treatment in this area is approached differently from other parts of the face. Confusion is common, not only about whether Botox is suitable, but also about safety, longevity, cost, and how it compares to alternatives such as dermal fillers or laser treatments. Having accurate information at the outset helps reduce risk and supports better conversations with practitioners.
Within the full guide, you will find an explanation of what causes lip lines, how Botox works in this area, what treatment typically involves, potential side effects, and how results are assessed over time. It also outlines when Botox may not be recommended, and why individual assessment matters.
Botox ForNeck Bands
You may think Botox is just reserved for the face, but did you know it can also be used to reduce neck wrinkles?
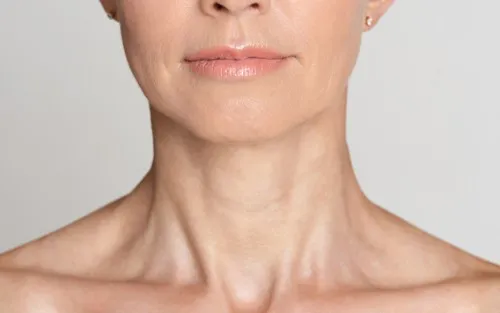
Botox for neck bands refers to the use of botulinum toxin injections to soften the appearance of vertical muscle bands that can become more noticeable in the neck over time. These bands are linked to muscle activity rather than skin quality alone, which is why this treatment is often discussed alongside, but not interchangeable with, skincare, devices, or surgery.
This topic matters because neck concerns are frequently misunderstood. Many people assume that visible bands are a sign of loose skin, when they are often driven by the underlying platysma muscle. That confusion can make it difficult to judge whether Botox is appropriate or whether another approach would be more suitable. As with any injectable treatment, understanding what is being treated and why supports safer, more realistic decision-making.
The full guide explains what neck bands are, how Botox is used in this area, how suitability is assessed in clinical practice, and what limitations to be aware of. It also covers practical considerations such as safety, treatment planning, and how outcomes are typically evaluated.
This page is designed to help you understand the role of Botox for neck bands and where it fits within wider treatment choices. Personalised advice will always depend on an in-person assessment, but informed context is a sensible starting point.
Botox For Gummy Smiles
If you think that too much of your gums are on show when you smile, Botox treatment for gummy smile could help.

Botox for a gummy smile refers to small, carefully placed injections that reduce how high the upper lip lifts when you smile. It is one of several options discussed when excess gum show becomes a concern, but it is also one of the most commonly misunderstood. This guide is designed to clarify what the treatment actually does, when it may be appropriate, and when other approaches are more relevant.
For many people, a gummy smile is not a single condition with a single solution. It can be influenced by muscle activity, lip structure, tooth position, or the relationship between the jaw and gums. Botox addresses only one of these factors: overactive upper lip muscles. Understanding that distinction matters, particularly if you are comparing treatments or trying to decide whether an injectable option makes sense for you at all.
The full guide looks at how Botox for a gummy smile is assessed in clinical practice, what a typical consultation should include, how the treatment works, how long effects tend to last, and the risks and limitations to be aware of. It also covers cost considerations and why practitioner judgement is particularly important with this treatment.
Botox ForExcessive Sweating (Hyperhidrosis)
Is your sweating out of control? If you're reapplying deodorant every couple of hours to your underarms with no avail, then you may be suffering from excessive sweating, also known by the medical term hyperhidrosis.

Excessive sweating, clinically known as hyperhidrosis, is a condition that goes beyond normal perspiration. It can affect specific areas such as the underarms, hands, feet, or face, often without clear triggers like heat or exercise. For many people, this raises practical questions about comfort, confidence, and the appropriateness of medical treatment.
Botulinum toxin injections are one option used in clinical practice to manage focal hyperhidrosis. They work by interrupting the nerve signals that stimulate sweat glands in the treated area. While the treatment is widely discussed, it is also frequently misunderstood, particularly regarding how it works, who it is suitable for, and what limitations to consider. Clear information matters here because treatment decisions often sit at the intersection of medical need and quality-of-life concerns.
Within the full guide, you’ll find clear explanations of how treatment is assessed in practice, which areas are commonly treated, what to expect in terms of duration and repeat appointments, and how safety and suitability are evaluated. The aim is to support informed, confident decision-making rather than quick conclusions.
Read the full guide: Botox For Excessive Sweating (Hyperhidrosis)
Botox ForMigraines
Interestingly, Botox injections can be used as a treatment for chronic migraine headaches and other headache-related conditions.

Botox for migraines is a medical use of botulinum toxin, prescribed for people with chronic migraine rather than occasional headaches. It is often discussed alongside cosmetic treatments, which can make it confusing to understand what it involves, who it is for, and how it is assessed in clinical practice. This page is designed to clarify those distinctions and explain where migraine treatment fits within regulated medical care.
For many people researching this option, the uncertainty lies in knowing when Botox is considered appropriate, how it differs from standard migraine medications, and whether it is something to explore through the NHS, private care, or both. Because treatment follows a specific medical protocol and is usually offered after other options have been tried, understanding the criteria and limitations matters. Clear information helps avoid unrealistic expectations and supports informed decisions about next steps.
This guide is most relevant if you experience frequent, long-term migraines that significantly affect daily life, or if you have been advised to look into Botox as part of a broader management plan. It may be less relevant for people with infrequent migraines or tension headaches, where other treatments are usually considered first.
Botox Foran Overactive Bladder
An overactive bladder is a frustrating medical condition that can hinder your life, treatment with Botox injections, has been proven to be highly effective in controlling urinary incontinence.

Botox for an overactive bladder is a medical use of botulinum toxin designed to help manage symptoms such as urgency, frequent urination, and leakage when other treatments have not been effective. While Botox is widely known in aesthetics, its use in bladder care is clinically distinct and often less well understood, which can make it difficult to know whether it is relevant to you.
Overactive bladder symptoms can significantly affect daily life, work, sleep, and confidence, yet many people delay exploring further options due to uncertainty or concern about what treatment involves. Botox is usually discussed only after conservative approaches, such as lifestyle changes or medication, have been tried. Understanding how and why it is used helps you assess whether it sits appropriately within your own treatment pathway, rather than viewing it as a last resort or a drastic step.
This guide is intended for adults who are researching next steps, comparing options, or preparing for a conversation with a GP, urology specialist, or continence clinic. It may be particularly relevant if symptoms persist despite first-line treatments. It may be less suitable if you are at an early stage of symptom management or have conditions that affect bladder emptying, which always require individual medical advice.
Botox Quick FAQs
Clear, Concise Answers to Common Queries
A simple, no-fluff roundup of frequent Botox questions to help you get the facts fast.

Can you have Botox if you are pregnant or breastfeeding?
Botox is generally not recommended during pregnancy or while breastfeeding because there is limited safety evidence in these groups. If you are trying to conceive, pregnant, or nursing, tell your prescriber before booking. They can talk you through safer timing and alternatives.
Can Botox affect your ability to drive or work afterwards?
Most people can drive and return to normal desk-based work straight after treatment. If you feel lightheaded, have a headache, or feel unwell, it is sensible to wait until you feel fully normal before driving. If your job involves strenuous physical work, your clinician may advise a short pause.
Is it safe to fly after Botox?
Flying is not usually a problem after Botox, as cabin pressure changes are not known to affect the product. The main consideration is that bruising or swelling can be more noticeable in the first couple of days, and you may not want to travel during that window. If you are flying the same day, follow your clinician’s aftercare advice carefully.
When can you wear make-up after Botox?
Many clinics advise waiting a few hours before applying makeup, mainly to reduce rubbing and pressure around injection sites. If you do apply makeup on the same day, use gentle patting rather than vigorous blending. Avoid facial massage tools until your clinician approves.
Can you exercise after Botox?
Strenuous exercise is often discouraged for the rest of the day, as increased blood flow and pressure can worsen swelling or bruising. Light walking is usually fine. If you are training for an event, ask for advice tailored to your routine and timing.
Does alcohol matter before or after Botox?
Alcohol can make bruising more likely because it can widen blood vessels and affect clotting in the short term. Some clinicians recommend avoiding alcohol for 24 hours before and after treatment, especially if you bruise easily. If you choose to drink, keep it modest and stay well hydrated.
What if one side looks slightly different after Botox?
Small differences can occur because faces are naturally asymmetrical and muscles respond at different rates. Results usually settle over 1–2 weeks, so early unevenness does not always mean the final outcome will be uneven. If you are concerned after the settling period, contact your provider for an assessment.
What happens if Botox does not work for you?
A lack of effect can be due to dosing, placement, muscle strength, or timing, rather than the product “failing”. In a small number of people, the body may respond less strongly over time. A qualified prescriber can review what was used and discuss adjustments or alternative treatments.
Can you build up resistance to Botox?
Resistance is considered uncommon but can happen, usually linked to repeated exposure over time and product factors. If results seem to reduce over multiple treatments, your clinician may consider changing technique, dose, or (where appropriate) switching to a different botulinum toxin type. This should always be guided by a prescriber.
Does Botox interact with vaccines or illness?
There is no routine requirement to avoid vaccines because of Botox, but timing can be sensible if you want to separate side effects (for example, headache or fatigue) from treatment effects. If you are unwell, have a fever, or have an active infection, it is usually best to postpone until you are fully recovered. Tell your clinician about recent vaccinations and any current illness.
Can you have Botox if you have a neurological or muscle condition?
Certain conditions that affect nerves or muscles may increase risk or make Botox unsuitable. Examples can include myasthenia gravis or other neuromuscular disorders. Always disclose your medical history and ask for a prescriber-led assessment rather than proceeding on assumptions.
What should you do if you think you are having a complication?
Mild redness, small bumps, or a headache can occur and often settle. Seek urgent medical advice if you have worrying symptoms such as difficulty breathing, swallowing problems, or rapidly worsening weakness. For non-urgent concerns (for example, drooping eyelid or unexpected asymmetry), contact your treating clinic promptly for guidance and review.

