
06
Sep
2012
It seems that no matter what the medicines regulations say or how many adjudications the Advertising Standards Authority (ASA) issues on the subject, the practice of promoting the prescription-only medicine of botulinum toxin, Botox® being one of the brand names, to the general public is still very much being done by UK clinics on a daily basis.
Smaller operators may be forgiven for not fully understanding the regulations in place given that people band the ‘botox’ word about with as much regard as we now think ‘hover’ is part of the English language, yet ignorance is never an excuse for flouting laws. Neither is claiming a ‘sheep mentality, in other words just because your competition down the road advertises Botox, doesn’t mean that it’s okay for you to do it – as mothers and teachers often say, “two wrongs don’t make a right”.
So you’d think some of the ‘big guys’ who have been around for years would and should know better and thus would not engage in this practice, wouldn’t you?
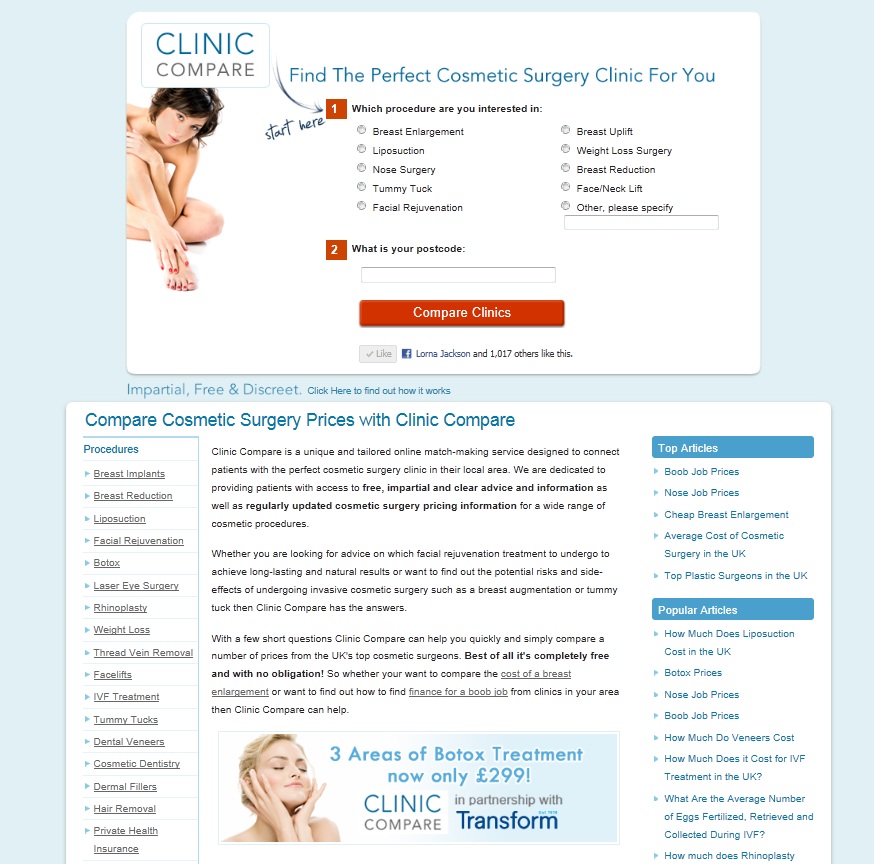
Well it seems not, as demonstrated in this banner advert (hosted on the cliniccompare.co.uk website this summer) for Botox treatments at the nationwide clinic chain for Transform Cosmetic Surgery Group. As a company established in 1974 they should be very well aware of the UK regulations on the advertising of medicines, specifically prescription-only ones.

From August 2012, the relevant UK legislation covering the advertising of medicines to the public is Part 14 of the Human Medicines Regulations 2012. This codifies and has replaced the Medicines (Advertising) Regulations 1994 and the Medicines (Monitoring of Advertising) Regulations 1994.
Now, this is not to say that clinics can’t advertise their services, they simply can’t mention prescription drugs by name (brand) or generic name. So for example, a clinic may promote the service provided, e.g. “treatment for lines and wrinkles”, as this is non-specific and may include various procedures which aren’t POMs such as dermal fillers, however advertising to the public must not mention product names such as “Botox” or “botulinum toxin”, which online banner adverts of this nature clearly do.
Perhaps the reason is simply that the lucrative gains from getting consumers through the door outweigh the small risk of being caught and told to cease your advertising strategy by the ASA and MHRA. In other words, no one with snarling teeth is there to really deter the practice so it persists. The more people get away with it, the more the good guys get annoyed and wonder whether they should ‘move to the dark side’ as well!
Hey, wait!
Before you go.....
Let's stay in touch, pop your details here and we'll send our editor's hand-picked updates on your fave subjects.