12
Jul
2010
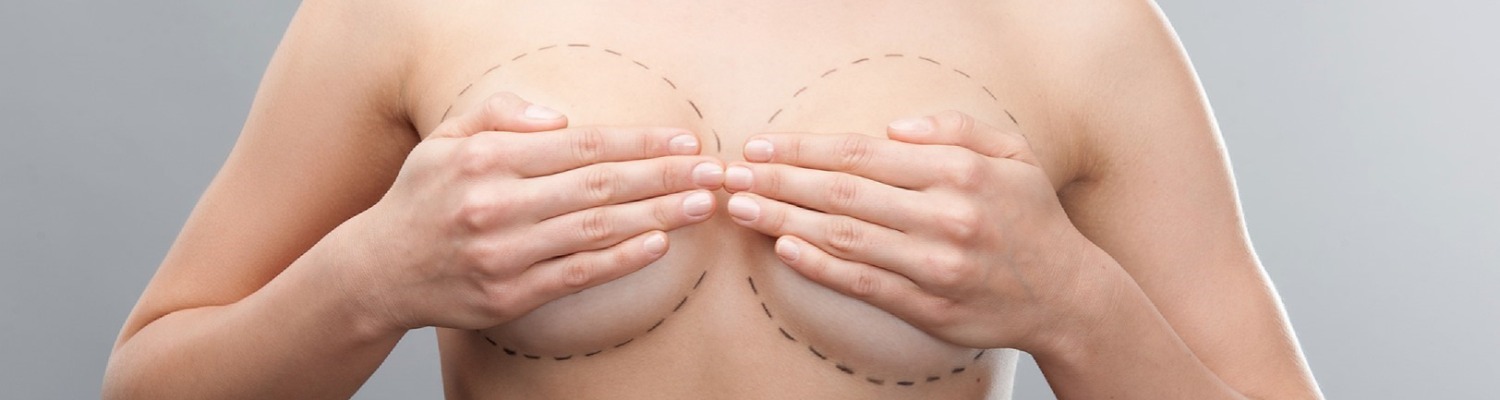
The British Association of Aesthetic Plastic Surgeons (BAAPS) has issued updated advice on Poly Implant Prothese (PIP) breast implants in which they urge all women fitted with the devices, now known to be faulty, to undergo an ultrasound scan in the next six months, to determine whether there is any rupture or weakening of the implant.
"This is certainly an unusual situation but so far there is no serious cause for alarm”; highlighted Consultant Plastic Surgeon and BAAPS President, Nigel Mercer.
“We recommend that women who've undergone breast augmentation contact their surgeons to find out what type of implant was used. If it's PIP they should have an ultrasound to establish whether there is any weakening or rupture. At present removal is recommended in these cases, but if there is one ruptured implant, the contra-lateral one should be taken out as well, as a preventative measure."
At the end of March 2010, the French medical regulatory authority (AFSSAPS) informed our own MHRA (Medicines and Healthcare products Regulatory Agency) that it had suspended the marketing, distribution, export and use of the silicone gel-filled PIP breast implants manufactured by a French company. The French authority recalled all of these devices at that point.

On inspecting the PIP manufacturing facility, based between Marseille and Nice in the South of France, the AFSSAPS found that breast implants made by the company since 2001 had been filled with a silicone gel with a composition different from that used to obtain their original European CE Mark approval, making this illegal.
This discovery cast much concern over the safety of the products and the MHRA advised all U.K. patients fitted with the PIP implants to consult their implanting surgeon if they had any concerns, whilst the French authorities investigated further.
One Cosmetic Surgeon that I spoke to said; “Anecdotal experience revealed a high rupture rate from surgeons and if you look on blog sites there are a lot of women who had problems with them, mostly relating to rupture and its consequences (i.e. capsule, asymmetry, lumps etc).”
Recent updates from the French Society of Plastic, Reconstructive and Aesthetic Surgery (SOFCPRE) have now revealed that from 2005 onwards the company also dispensed with a protective barrier which normally surrounds an implant to offer strength and reduce the migration risk of the gel. In addition, it became clear to SOFCPRE, on contacting the actual gel manufacturer of the substitute silicone gel used by the implant manufacturers that there was a lack of clinical data in relation to its possible reaction with the human body, as the substance was intended for use in mattresses and they were unaware of its eventual use in breast implants!
Despite proving unpopular with the majority of independent surgeons, who tended to prefer more expensive and better quality implant products which provided a lifetime guarantee against rupture for their patients, it has been estimated that roughly 50,000 women in the UK could have these PIP breast implants, as they were the product of choice for some clinics, particularly those larger cosmetic surgery chains where it was the bottom line which drove choice.
"This situation is clearly not the fault of the surgeon, who acted in good faith - it would be similar to blaming a dealership for a faulty car."; concluded Nigel Mercer.
A spokesperson for Clover Leaf Products, the former distributor of PIP implants in the UK said on 23rd June; “All medical devices are regulated in the UK by the MHRA who issued a device alert on the 31st March 2010. The action for surgeons on this alert was as follows: ‘Await further advice from the MHRA regarding clinical management of patients implanted with these devices’. To our knowledge, this is still the case today and we are awaiting further information.”
Following delays from AFSSAPS who have stated that the results from their own tests are now expected in September, the UK’s MHRA has decided to conduct its own toxicology tests into the gel contained within the PIP implants, which they state should be completed in mid-August.
If you want to read more, the experts at Consulting Room really know what they're talking about and have put together Breast Surgery and Breast Augmentation FAQs just for you.
If you have more questions, you can use the Breast Surgery and Breast Augmentation questions feature to talk to our panel of trained medical experts.
If you're keen to get started with any of these treatments right away then you're in luck - those clever folks also have a list of trusted, accredited Breast Surgery and Breast Augmentation clinics in your area.
Did you know that the UK is the only country in the world with an unregulated aesthetic industry?
The recent documentary by Olivia Attwood shows how confusing it can be to choose who and where to get your aesthetic treatments from, and how safe they are...
2023 marks a momentous milestone as Consulting Room celebrates 20 years of excellence!
Hey, wait!
Before you go.....
Let's stay in touch, pop your details here and we'll send our editor's hand-picked updates on your fave subjects.